A core tenet of evolutionary biology, ingrained in mainstream thinking for more than a century, is that genetic mutations occur randomly, without respect to the needs of the organism. A recent paper, by a team led by Daniel Melamed of the University of Haifa in Israel, has challenged this paradigm.1 The study, published in the Proceedings of the National Academy of Sciences, reveals that a mutation in the human APOL1 gene arises with a greater frequency when it serves an advantage of preventing disease.
Previous Evidence from the Sickle-Cell Mutation
In a previous study by the same team, a new sensitive detection method was developed and applied to the HbS mutation which confers protection from malaria in individuals possessing one copy of the mutation (heterozygotes) but results in sickle-cell anemia in individuals carrying two copies of the mutation (homozygotes). The results of this study were published in 2022.2
The APOL1 Mutation
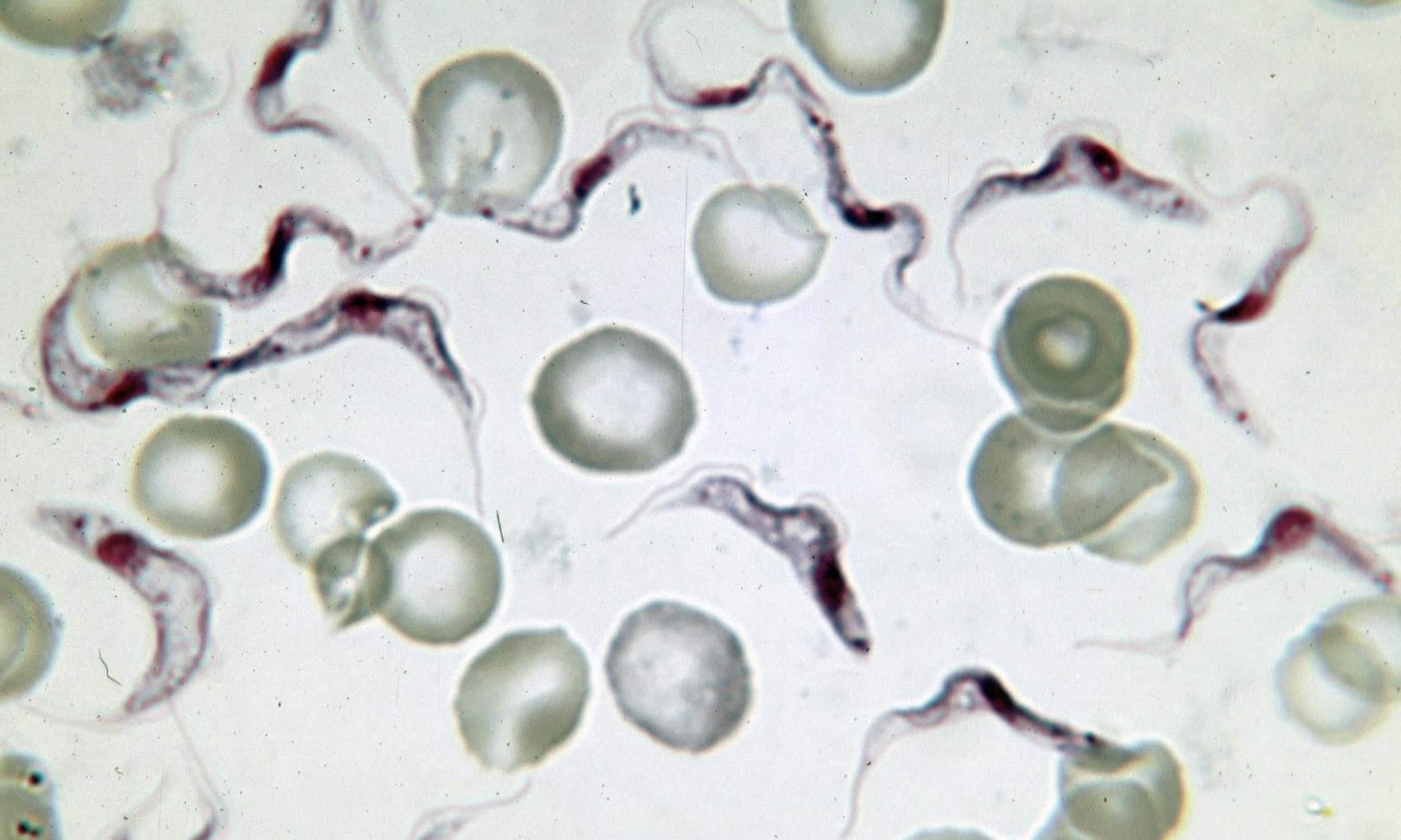
In this newer study, the researchers examined the emergence of a mutation in the human APOL1 gene that confers protection from African sleeping sickness, a disease caused by the parasite Trypanosoma brucei (pictured at the top). The authors extracted sperm DNA from healthy donors in Africa and Europe, sampling as many as 291 million African genomes and 323 million European genomes. Since each sperm cell represents one haploid germline genome, counting mutations in sperm yields a direct estimate of the frequency with which mutations arise. The mutation in question does not affect sperm viability, and thus its likelihood of appearing is equivalent to its frequency in sperm.
The researchers employed a method called Mutation Enrichment followed by Maximum Depth Sequencing (MEMDS) to detect mutations. This technique uses a restriction enzyme (HindIII) to cut and remove wild-type DNA, leaving behind the mutation DNA, where the restriction site is disrupted. They compared treated and untreated samples to determine how many original genomes were scanned and how much wild-type DNA was removed, thereby determining the mutation frequency in the original sample.
On the hypothesis that the APOL1 mutation arises randomly, it should emerge at a similar frequency in all populations but only become fixed in sub-Saharan African populations where there is a selective pressure for it to increase in frequency in the population. If, conversely, it arises nonrandomly, it may in fact arise more frequently in the regions where it confers an advantage.
The results indicated that the mutation emerged significantly more frequently among sub-Saharan African populations where sleeping sickness is endemic than in European populations. These data are consistent with the hypothesis that the APOL1 mutation arises nonrandomly.
Pre-Programmed Adaptive Capacity
These data suggest that the mutation process is biased in ways that are biologically meaningful. If these mechanisms reflect a phenomenon that can be generalized, it would represent a significant overhaul of the standard assumption of the randomness of genetic mutations. On the other hand, that there exists a pre-programmed adaptive capacity — equipping organisms to anticipate and react to environmental challenges — is precisely the sort of thing that one might expect to see on the hypothesis that living organisms are the product of intentional engineering.
Notes
- Melamed D, Shemer R, Bolotin E, Yakass MB, Fink-Barkai D, Hiadzi EK, Skorecki KL, Livnat A. De novo rates of a Trypanosoma-resistant mutation in two human populations. Proc Natl Acad Sci U S A. 2025 Sep 2;122(35):e2424538122. doi: 10.1073/pnas.2424538122. Epub 2025 Aug 25. PMID: 40854136; PMCID: PMC12415191.
- Melamed D, Nov Y, Malik A, Yakass MB, Bolotin E, Shemer R, Hiadzi EK, Skorecki KL, Livnat A. De novo mutation rates at the single-mutation resolution in a human HBB gene region associated with adaptation and genetic disease. Genome Res. 2022 Mar;32(3):488-498. doi: 10.1101/gr.276103.121. Epub 2022 Jan 14. PMID: 35031571; PMCID: PMC8896469.